CAZypedia needs your help! We have many unassigned GH, PL, CE, AA, GT, and CBM pages in need of Authors and Responsible Curators.
Scientists at all career stages, including students, are welcome to contribute to CAZypedia. Read more here, and in the 10th anniversary article in Glycobiology.
New to the CAZy classification? Read this first.
*
Consider attending the 15th Carbohydrate Bioengineering Meeting in Ghent, 5-8 May 2024.
Glycoside Hydrolase Family 23
This page has been approved by the Responsible Curator as essentially complete. CAZypedia is a living document, so further improvement of this page is still possible. If you would like to suggest an addition or correction, please contact the page's Responsible Curator directly by e-mail.
| Glycoside Hydrolase Family GH23 | |
| Clan | none, α+β "lysozyme fold" |
| Mechanism | retaining/inverting |
| Active site residues | known |
| CAZy DB link | |
| http://www.cazy.org/GH23.html | |
Substrate specificities
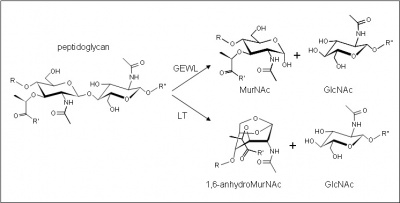
The glycoside hydrolases of this family are lytic transglycosylases (also referred to as peptidoglycan lyases) of both bacterial and bacteriophage origin, and family G lysozymes (EC 3.2.1.17; muramidase, peptidoglycan N-acetylmuramoylhydrolase, 1,4-β-N-acetylmuramidase, N-acetylmuramoylhydrolase) of eukaryotic origin. Both of these enzymes are active on peptidoglycan, but only the lysozymes are active on chitin and chitooligosaccharides. No other activities have been observed.
The lytic transglycosylases of GH23 constitute Family 1 of the organizational scheme of Blackburn and Clarke [1]. This family has been subdivided into five subfamilies (1A-1E) with Escherichia coli soluble lytic transglycosylase 70 (Slt70), membrane-bound lytic transglycosylase C (MltC), MltE, MltD, and MltF (formerly, YfhD) serving as the prototypes for families 1A, 1B, 1C, 1D, and 1E, respectively.
Kinetics and Mechanism
The enzymes of this family cleave the β-1,4-linkage between N-acetylmuramyl and N-acetylglucosaminyl residues in peptidoglycan (Figure 1). Only the lysozymes of this family are capable of releasing N-acetyl-d-glucosamine residues from chitodextrins, and neither catalyze transglycosylation reactions. The stereochemistry of the reaction catalysed by the family G lysozymes has been determined experimentally as inverting [2]. On the other hand, the lytic transglycosidases, strictly speaking, are retaining enzymes. However, unlike lysozyme, they are not hydrolases but rather catalyse an intramolecular transglycosylation to the C-6 hydroxyl group of the muramyl residue leading to the generation of a terminal 1,6-anhydromuramic acid product that is an acetal, and not a hemiacetal [3]. The lytic transglycosylases require the peptide side chains in peptidoglycan for activity, accounting for their inactivity against chitin or chitooligosaccharides [4]. No detailed analyses involving both steady-state and pre-steady state kinetic studies have been reported.
Catalytic Residues
Until recently, the family GH23 enzymes were thought to have only a single catalytic residue at their active centre. The identity of the catalytic general acid/base residue of the lysozymes was first inferred by X-ray crystallography of goose egg-white lysozyme (GEWL) as Glu73 [5, 6]. Likewise, analysis of the crystal structure of E. coli Slt70 identified Glu as the lone catalytic residue[7]. Replacement of each respective residue results in loss of catalytic activity [2, 8]. The mechanism of action of the family GH23 enzymes has yet to be proven experimentally, but examination of crystal structures and theoretical considerations have led to separate proposals for the two classes of enzymes.
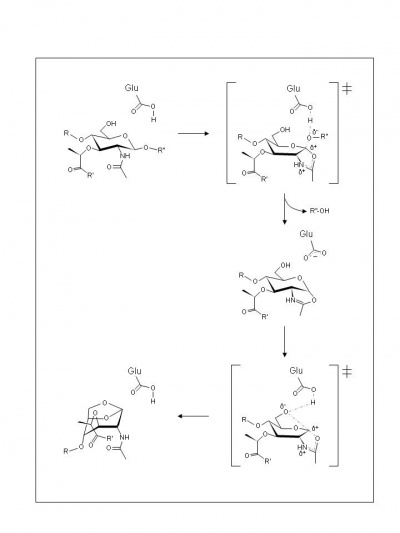
With the lytic transglycosylases, there is still no evidence for a second catalytic residue at their active sites. Hence, based on the complexes formed with 1,6-anhydromuropeptide [9] or bulgecin [10], a two-step mechanism that uses neighboring group participation (also termed substrate assisted catalysis), analogous to the family GH18 chitinases and chitobiases and family GH20 N-acetyl-β-hexosaminidases, has been invoked. Thus, in the first step the Glu73 residue is proposed to serve initially as a general acid to donate a proton to the glycosidic oxygen of the linkage (Figure 2). At the same time anchimeric assistance by the MurNAc 2-acetamido group results in the formation an oxazolinium ion intermediate. In the second step Glu73 acts as a general base to abstract the proton from the C-6 hydroxyl leading to its nucleophilic attack on the anomeric centre and the formation of 1,6-anhydromuramic acid product. The transition states leading to and from the intermediate possess oxocarbenium ion character (Figure 2).
For the g-type lysozymes, recent studies support a typical inverting mechanism of action [11, 12] involving general acid and general base residues. It was proposed that two catalytic general base residues, a primary and a secondary residue, position a catalytic water molecule and abstract a proton to effect substrate hydrolysis by the single displacement mechanism. With GEWL, Asp97 and Asp86, respectively, are proposed to serve this function while these would be represented by the conserved Asp101 and Asp90 residues in the g-type lysozyme from Atlantic cod fish. Indeed, the double replacement of Asp101 and Asp90 in the cod lysozyme results in over a 300-fold decrease in activity [11].
Three-dimensional structures
Three-dimensional structures are available for several Family GH23 enzymes, the first solved being that of GEWL [6, 13]. The catalytic domain of each enzyme possesses the well characterized α+β "lysozyme fold" for avian lysozymes. However, there are distinct structural differences between the two classes of enzymes. Most notably, the environment of the active site in lytic transglycosylases, particularly around the catalytic acid/base, is more hydrophobic compared to that of GEWL. This distinction may account for the difference in the reaction mechanisms of the two enzymes.
Family Firsts
- First identification of lytic transglycosylase
- Soluble lytic transglycosylase 70 [3].
- First catalytic nucleophile identification
- For g-type lysozymes [12]; Not applicable for lytic transglycosylases.
- First general acid/base residue identification
- Inferred by X-ray crystallography of goose egg-white lysozyme [6].
- First 3-D structure
- Goose egg-white lysozyme [13].
References
- Blackburn NT and Clarke AJ. (2001). Identification of four families of peptidoglycan lytic transglycosylases. J Mol Evol. 2001;52(1):78-84. DOI:10.1007/s002390010136 |
- Kuroki R, Weaver LH, and Matthews BW. (1999). Structural basis of the conversion of T4 lysozyme into a transglycosidase by reengineering the active site. Proc Natl Acad Sci U S A. 1999;96(16):8949-54. DOI:10.1073/pnas.96.16.8949 |
- Höltje JV, Mirelman D, Sharon N, and Schwarz U. (1975). Novel type of murein transglycosylase in Escherichia coli. J Bacteriol. 1975;124(3):1067-76. DOI:10.1128/jb.124.3.1067-1076.1975 |
- Romeis T, Vollmer W, and Höltje JV. (1993). Characterization of three different lytic transglycosylases in Escherichia coli. FEMS Microbiol Lett. 1993;111(2-3):141-6. DOI:10.1111/j.1574-6968.1993.tb06376.x |
- Weaver LH, Grütter MG, and Matthews BW. (1995). The refined structures of goose lysozyme and its complex with a bound trisaccharide show that the "goose-type" lysozymes lack a catalytic aspartate residue. J Mol Biol. 1995;245(1):54-68. DOI:10.1016/s0022-2836(95)80038-7 |
- Weaver LH, Grütter MG, Remington SJ, Gray TM, Isaacs NW, and Matthews BW. (1984-1985). Comparison of goose-type, chicken-type, and phage-type lysozymes illustrates the changes that occur in both amino acid sequence and three-dimensional structure during evolution. J Mol Evol. 1984-1985;21(2):97-111. DOI:10.1007/BF02100084 |
- van Asselt EJ, Thunnissen AM, and Dijkstra BW. (1999). High resolution crystal structures of the Escherichia coli lytic transglycosylase Slt70 and its complex with a peptidoglycan fragment. J Mol Biol. 1999;291(4):877-98. DOI:10.1006/jmbi.1999.3013 |
- van Asselt EJ, Dijkstra AJ, Kalk KH, Takacs B, Keck W, and Dijkstra BW. (1999). Crystal structure of Escherichia coli lytic transglycosylase Slt35 reveals a lysozyme-like catalytic domain with an EF-hand. Structure. 1999;7(10):1167-80. DOI:10.1016/s0969-2126(00)80051-9 |
- Thunnissen AM, Isaacs NW, and Dijkstra BW. (1995). The catalytic domain of a bacterial lytic transglycosylase defines a novel class of lysozymes. Proteins. 1995;22(3):245-58. DOI:10.1002/prot.340220305 |
- Thunnissen AM, Rozeboom HJ, Kalk KH, and Dijkstra BW. (1995). Structure of the 70-kDa soluble lytic transglycosylase complexed with bulgecin A. Implications for the enzymatic mechanism. Biochemistry. 1995;34(39):12729-37. DOI:10.1021/bi00039a032 |
- Helland R, Larsen RL, Finstad S, Kyomuhendo P, and Larsen AN. (2009). Crystal structures of g-type lysozyme from Atlantic cod shed new light on substrate binding and the catalytic mechanism. Cell Mol Life Sci. 2009;66(15):2585-98. DOI:10.1007/s00018-009-0063-x |
- Hirakawa H, Ochi A, Kawahara Y, Kawamura S, Torikata T, and Kuhara S. (2008). Catalytic reaction mechanism of goose egg-white lysozyme by molecular modelling of enzyme-substrate complex. J Biochem. 2008;144(6):753-61. DOI:10.1093/jb/mvn133 |
- Grütter MG, Weaver LH, and Matthews BW. (1983). Goose lysozyme structure: an evolutionary link between hen and bacteriophage lysozymes?. Nature. 1983;303(5920):828-31. DOI:10.1038/303828a0 |