CAZypedia needs your help! We have many unassigned GH, PL, CE, AA, GT, and CBM pages in need of Authors and Responsible Curators.
Scientists at all career stages, including students, are welcome to contribute to CAZypedia. Read more here, and in the 10th anniversary article in Glycobiology.
New to the CAZy classification? Read this first.
*
Consider attending the 15th Carbohydrate Bioengineering Meeting in Ghent, 5-8 May 2024.
Glycoside Hydrolase Family 103
This page has been approved by the Responsible Curator as essentially complete. CAZypedia is a living document, so further improvement of this page is still possible. If you would like to suggest an addition or correction, please contact the page's Responsible Curator directly by e-mail.
- Author: ^^^Anthony Clarke^^^
- Responsible Curator: ^^^Anthony Clarke^^^
| Glycoside Hydrolase Family GHnn | |
| Clan | None
α+β "lysozyme fold" |
| Mechanism | retaining |
| Active site residues | known |
| CAZy DB link | |
| http://www.cazy.org/fam/GHnn.html | |
Substrate specificities
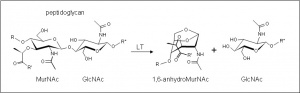
The glycoside hydrolases of this family are lytic transglyosylases (also referred to as peptidoglycan lyases) of bacterial origin and they constitute family 3 of the classification scheme of Blackburn and Clarke [1]. The prototype for this family is membrane-bound lytic transglycosylase B (MltB) from Escherichia coli [2]. These enzymes cleave the β-1,4 linkage between N-acetylmuramoyl and N-acetylglucosaminyl residues in peptidoglycan (Figure 1), No other activities have been observed.
Kinetics and Mechanism
The lytic transglycosidases, strictly speaking, are retaining enzymes. However, they are not hydrolases but rather catalyse an intramolecular glycosyl transferase reaction onto the C-6 hydroxyl group of the muramoyl residue leading to the generation of a terminal 1,6-anhdyromuramoyl product (Figure 1) thus lacking a reducing end [3]. No detailed analyses involving both steady state and pre-steady state kinetic studies have been reported, but the Michaelis Menten (KM and Vmax) parameters have been estimated for Pseudomonas aeruginosa MltB acting on insoluble peptidoglycan sacculi [4].
Catalytic Residues
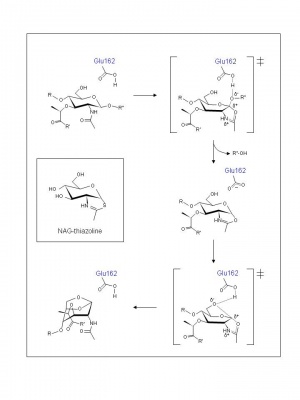
As with other lytic transglycosylases (families GH23, GH102, and GH104), the GH103 enzymes are thought to possess a single catalytic acid/base residue. This residue has been identified as Glu162 in MltB from both E. coli and P. aeruginosa and, indeed, its replacement abolishes catalytic activity [4, 5]. The mechanism of action of family GH103 enzymes has been investigated the most compared to the lytic transglycosylases of the other families (GH23,GH102, and GH104).
Examination of crystal structures of E. coli Slt35 (a soluble proteolytic derivative of MltB) and theoretical considerations led to the proposal of a mechanism that accommodates a single catalytic at its active site. Thus, based on the complexes formed with murodipeptide, chitobiose, and the inhibitor bulgecin, a substrate-assisted mechanism has been invoked analogous to the family GH18 chitinases and chitobiases, family GH20 N-acetyl-β-hexosaminidases, and family GH23 lytic transglycosylases [6]. Thus, the catalytic Glu162 is proposed to serve initially as an acid catalyst to donate a proton to the glycosidic oxygen of the linkage to be cleaved leading to the formation of an intermediate with oxocarbenium ion character (Figure 2). In the absence of an anion/nucleophile in close proximity to stabilize this oxocarbenium intermediate, the lytic transglycosylases would employ anchimeric assistance of the MurNAc 2-acetamido group resulting in the formation an oxazolinium ion intermediate. This would be followed by abstraction of the C-6 hydroxyl proton of the oxazolinium species involving Glu162 which now serves as the base catalyst leading to nucleophilic attack and the formation of 1,6-anhydromuramic acid product. The β-hexosaminidase inhibitor NAG-thiazoline (Figure 2) was found to inhibit P. aeruginosa MltB thus supporting the proposal for the formation of an oxazolinium ion intermediate [7], and the results of a site-directed mutagenesis study suggest that Ser216 orients the N-acetyl group on MurNAc at the -1 subsite of MltB for its participation in a substrate-assisted mechanism of action [8].
Three-dimensional structures
Three-dimensional structures are available for several family GH103 enzymes, the first solved being that of E. coli MltB (Slt35) [5]. The catalytic domain of the enyzmes possesses the well characterized α+β "lysozyme fold."
Family Firsts
- First identification of lytic transglycosylase
- MltB from E. coli [2].
- First catalytic nucleophile identification
- Not applicable for lytic transglycosylases.
- First general acid/base residue identification
- Inferred by X-ray crystallography of E. coli MltB [5].
- First 3-D structure
- E. coli MltB [5].
- First identification as a lipoprotein
- E. coli MltB [9].
- First identification of localization to outer membrane
- E. coli MltB [9].
- Frist demonstration of molecular interactions between GH103 enzymes and penicillin-binding proteins
- E. coli MltB [10].
References
- Blackburn NT and Clarke AJ. (2001). Identification of four families of peptidoglycan lytic transglycosylases. J Mol Evol. 2001;52(1):78-84. DOI:10.1007/s002390010136 |
- Engel H, Smink AJ, van Wijngaarden L, and Keck W. (1992). Murein-metabolizing enzymes from Escherichia coli: existence of a second lytic transglycosylase. J Bacteriol. 1992;174(20):6394-403. DOI:10.1128/jb.174.20.6394-6403.1992 |
- Höltje JV, Mirelman D, Sharon N, and Schwarz U. (1975). Novel type of murein transglycosylase in Escherichia coli. J Bacteriol. 1975;124(3):1067-76. DOI:10.1128/jb.124.3.1067-1076.1975 |
- Blackburn NT and Clarke AJ. (2002). Characterization of soluble and membrane-bound family 3 lytic transglycosylases from Pseudomonas aeruginosa. Biochemistry. 2002;41(3):1001-13. DOI:10.1021/bi011833k |
- van Asselt EJ, Dijkstra AJ, Kalk KH, Takacs B, Keck W, and Dijkstra BW. (1999). Crystal structure of Escherichia coli lytic transglycosylase Slt35 reveals a lysozyme-like catalytic domain with an EF-hand. Structure. 1999;7(10):1167-80. DOI:10.1016/s0969-2126(00)80051-9 |
- van Asselt EJ, Kalk KH, and Dijkstra BW. (2000). Crystallographic studies of the interactions of Escherichia coli lytic transglycosylase Slt35 with peptidoglycan. Biochemistry. 2000;39(8):1924-34. DOI:10.1021/bi992161p |
- Reid CW, Blackburn NT, Legaree BA, Auzanneau FI, and Clarke AJ. (2004). Inhibition of membrane-bound lytic transglycosylase B by NAG-thiazoline. FEBS Lett. 2004;574(1-3):73-9. DOI:10.1016/j.febslet.2004.08.006 |
- Reid CW, Legaree BA, and Clarke AJ. (2007). Role of Ser216 in the mechanism of action of membrane-bound lytic transglycosylase B: further evidence for substrate-assisted catalysis. FEBS Lett. 2007;581(25):4988-92. DOI:10.1016/j.febslet.2007.09.037 |
- Ehlert K, Höltje JV, and Templin MF. (1995). Cloning and expression of a murein hydrolase lipoprotein from Escherichia coli. Mol Microbiol. 1995;16(4):761-8. DOI:10.1111/j.1365-2958.1995.tb02437.x |
- von Rechenberg M, Ursinus A, and Höltje JV. (1996). Affinity chromatography as a means to study multienzyme complexes involved in murein synthesis. Microb Drug Resist. 1996;2(1):155-7. DOI:10.1089/mdr.1996.2.155 |