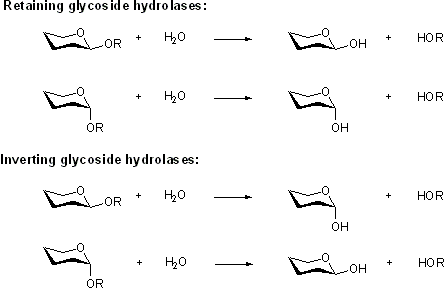CAZypedia needs your help! We have many unassigned GH, PL, CE, AA, GT, and CBM pages in need of Authors and Responsible Curators.
Scientists at all career stages, including students, are welcome to contribute to CAZypedia. Read more here, and in the 10th anniversary article in Glycobiology.
New to the CAZy classification? Read this first.
*
Consider attending the 15th Carbohydrate Bioengineering Meeting in Ghent, 5-8 May 2024.
Difference between revisions of "Glycoside Hydrolases"
| Line 45: | Line 45: | ||
[[Image:Retaining_glycosidase_mechanism_1.png|centre]] | [[Image:Retaining_glycosidase_mechanism_1.png|centre]] | ||
| − | Enzymes of glycoside hydrolase families [[Glycoside hydrolase family 18|18]], [[Glycoside hydrolase family 20|20]], [[Glycoside hydrolase family 56|56]] and [[Glycoside hydrolase family 84|84]] hydrolyse substrates containing an ''N''-acetyl (acetamido) group at the 2-position. These enzymes have no catalytic nucleophile: rather they utilize a mechanism in which the 2-acetamido group acts as an intramolecular nucleophile. Neighboring group participation by the 2-acdetamido group leads to formation of an oxazoline (or more strictly an oxazolinium ion) intermediate. This mechanism was deduced from X-ray structures of complexes of chitinases with natural inhibitors, from the potent inhibition afforded by a stable thiazoline analogue of the oxazoline, and from detailed mechanistic analyses using substrates of modified reactivity. Typically, a stabilizing residue (a carboxylate) stabilizes the charge development in the [[transition state]]. Not all enzymes that cleave substrates possessing a 2-acetamido group utilize a neighboring groups participation mechanism; enzyme of glycoside hydrolase families [[Glycoside hydrolase family 3|3]], [[Glycoside hydrolase family 19|19]] and [[Glycoside hydrolase family 85|85]] utilize a classical retaining mechanism with an enzymic nucleophile. Other hexosaminidases utilize an inverting mechanism. | + | Enzymes of glycoside hydrolase families [[Glycoside hydrolase family 18|18]], [[Glycoside hydrolase family 20|20]], [[Glycoside hydrolase family 56|56]] and [[Glycoside hydrolase family 84|84]] hydrolyse substrates containing an ''N''-acetyl (acetamido) group at the 2-position. These enzymes have no catalytic nucleophile: rather they utilize a mechanism in which the 2-acetamido group acts as an intramolecular nucleophile. Neighboring group participation by the 2-acdetamido group leads to formation of an oxazoline (or more strictly an oxazolinium ion) intermediate. This mechanism was deduced from X-ray structures of complexes of chitinases with natural inhibitors <cite>6</cite>, from the potent inhibition afforded by a stable thiazoline analogue of the oxazoline <cite>7</cite>, and from detailed mechanistic analyses using substrates of modified reactivity. Typically, a stabilizing residue (a carboxylate) stabilizes the charge development in the [[transition state]]. Not all enzymes that cleave substrates possessing a 2-acetamido group utilize a neighboring groups participation mechanism; enzyme of glycoside hydrolase families [[Glycoside hydrolase family 3|3]], [[Glycoside hydrolase family 19|19]] and [[Glycoside hydrolase family 85|85]] utilize a classical retaining mechanism with an enzymic nucleophile. Other hexosaminidases utilize an inverting mechanism. |
[[Image:Hex_neighboring_mechanism.png|centre]] | [[Image:Hex_neighboring_mechanism.png|centre]] | ||
| Line 56: | Line 56: | ||
#4 pmid=1618761 | #4 pmid=1618761 | ||
#5 pmid=7624375 | #5 pmid=7624375 | ||
| + | |||
| + | #6 pmid= 7495789 | ||
| + | |||
| + | #7 pmid= 11124970 | ||
</biblio> | </biblio> | ||
[[Category:Definitions and explanations]] | [[Category:Definitions and explanations]] | ||
Revision as of 03:10, 20 July 2009
- Author: Stephen Withers
- Responsible Editor: Spencer Williams
Overview
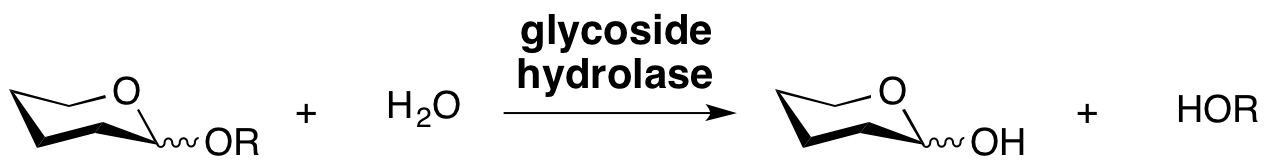
Glycoside hydrolases are enzymes that catalyze the hydrolysis of the glycosidic linkage of glycosides, leading to the formation of a sugar hemiacetal or hemiketal and the corresponding free aglycon. Glycoside hydrolases are also referred to as glycosidases, and sometimes also as glycosyl hydrolases. Glycoside hydrolases can catalyze the hydrolysis of O-, N- and S-linked glycosides.
Classification
Glycoside hydrolases can be classified in many different ways. The following paragraphs list several different ways, the utility of which depends on the context in which the classification is made and used.
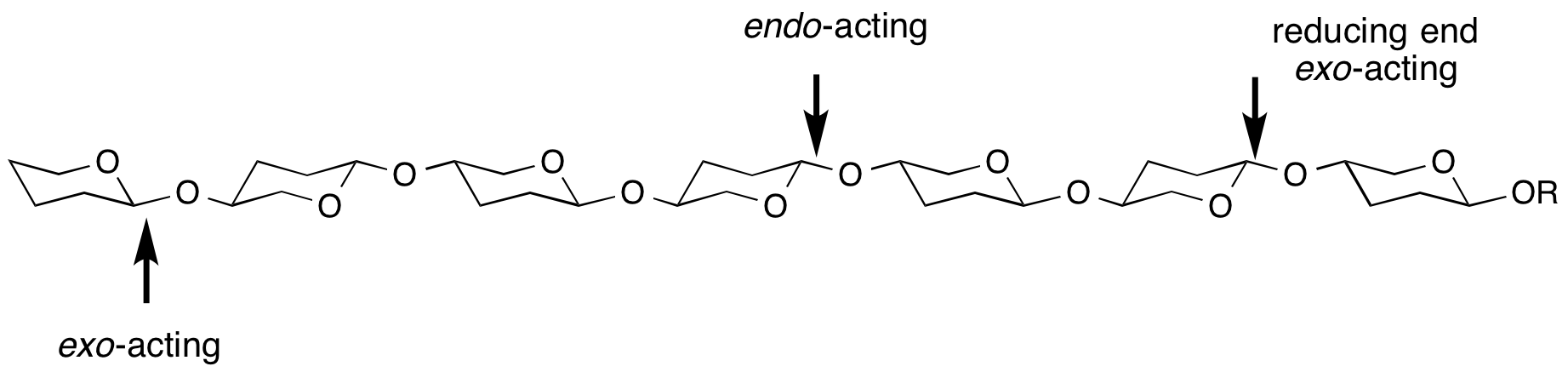
Endo/exo
exo- and endo- refers to the ability of a glycoside hydrolase to cleave a substrate at the end (most frequently, but not always the non-reducing end) or within the middle of a chain. For example, most cellulases are endo-acting, whereas LacZ β-galactosidase from E. coli is exo-acting.
Enzyme Commission (EC) number
EC numbers are codes representing the Enzyme Commission number. This is a numerical classification scheme for enzymes, based on the chemical reactions they catalyze. Every EC number is associated with a recommended name for the respective enzyme. EC numbers do not specify enzymes, but enzyme-catalyzed reactions. If different enzymes (for instance from different organisms) catalyze the same reaction, then they receive the same EC number. A necessary consequence of the EC classification scheme is that codes can be applied only to enzymes for which a function has been biochemically identified. Additionally, certain enzymes can catalyze reactions that fall in more than one class. These enzymes must bear more than one EC number.
Mechanistic classification
Two reaction mechanisms are most commonly found for the retaining and inverting enzymes, as first outlined by Koshland and as described below.[1] However several interesting variations on these mechanisms have been found, and one fundamentally different mechanism, catalyzed by an NADH cofactor, has been discovered in recent years, as discussed below.
Sequence classification
Sequence classification methods require knowledge of at least part of the amino acid sequence for an enzyme. Algorithmic methods are then used to compare sequences, and in the case of the glycoside hydrolases, this has allowed their classification into more than 100 families. Each family (GH family) contains proteins that are related by sequence, and by corollary, fold. An obvious shortcoming of sequence-based classifications is that they can only be applied to enzymes for which sequence information is available. On the other hand sequence-based classification schemes allow classification of proteins for which no biochemical evidence has been obtained such as the thousands of uncharacterized glycosidase-related sequences that orginate from genome sequencing efforts worldwide. This allows a number of useful predictions to be made since it has long been noted that the catalytic machinery and molecular mechanism is conserved for the vast majority of the glycosidase families [1] as well as the geometry around the glycosidic bond (irrespective of naming conventions) [2]. As such sequence based classification methods are rather different (and in many ways complimentary) to the EC method discussed above.
Mechanism
Two reaction mechanisms are most commonly found for the retaining and inverting enzymes, as first outlined by Koshland and as described below [3]. However several interesting variations on these mechanisms have been found, and one fundamentally different mechanism, catalyzed by an NADH cofactor, has been discovered in recent years.
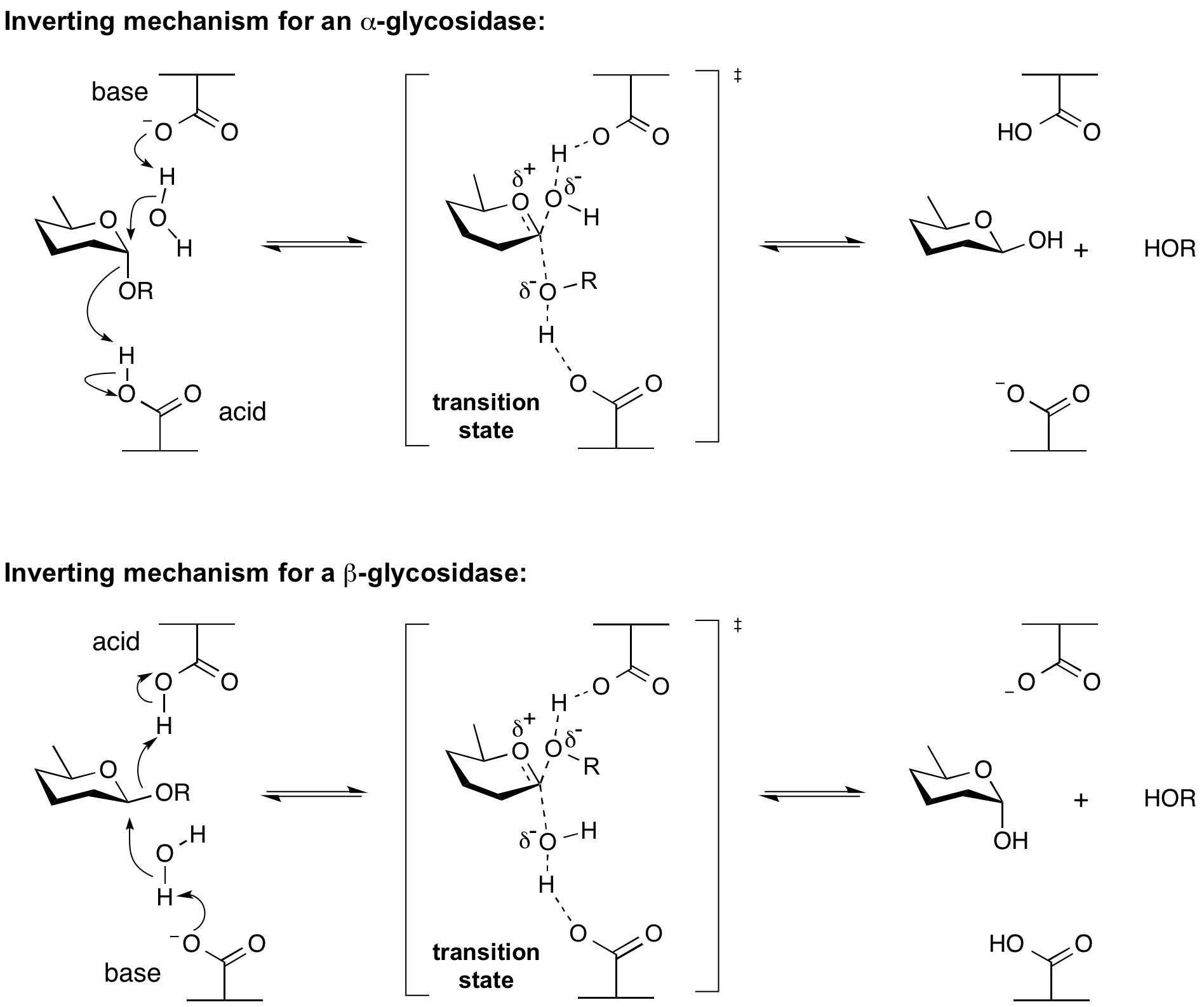
Inverting glycoside hydrolases
Hydrolysis of a glycoside with net inversion of anomeric configuration is generally achieved via a one step, single-displacement mechanism involving oxocarbenium ion-like transition states, as shown below. The reaction occurs with acid/base assistance from two amino acid side chains, normally glutamic or aspartic acids, that are typically located 6-11 A apart[4].
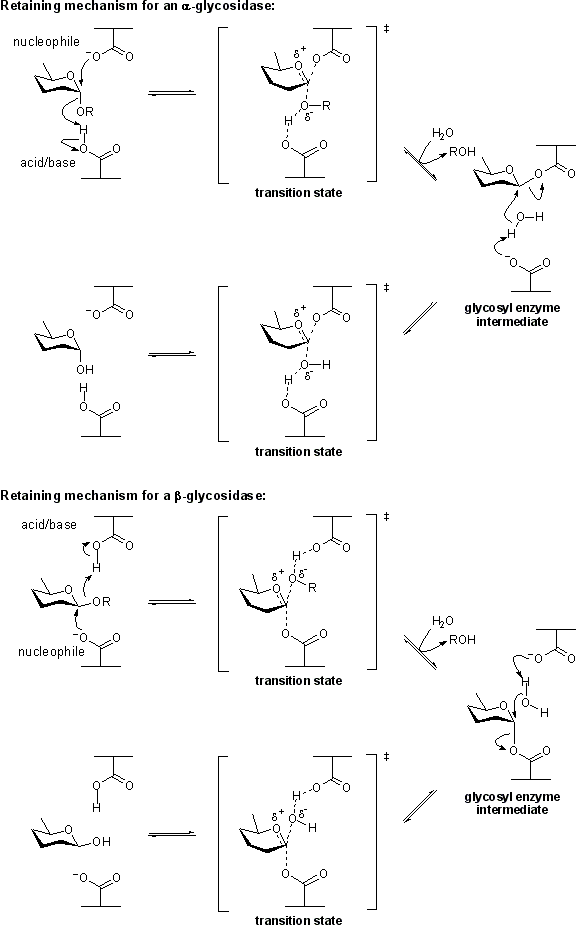
Retaining glycoside hydrolases
Hydrolysis with net retention of configuration is most commonly achieved via a two step, double-displacement mechanism involving a covalent glycosyl-enzyme intermediate, as is shown in Figure 3. Each step passes through an oxocarbenium ion-like transition state. Reaction occurs with acid/base assistance provided by two amino acid side chains, typically glutamate or aspartate, located 5.5 A apart. In the first step (often called the glycosylation step), one residue plays the role of a nucleophile, attacking the anomeric centre to displace the aglycon and form a glycosyl enzyme intermediate. At the same time the other residue functions as an acid catalyst and protonates the glycosidic oxygen as the bond cleaves. In the second step (known as the deglycosylation step), the glycosyl enzyme is hydrolyzed by water, with the other residue now acting as a base catalyst deprotonating the water molecule as it attacks. The pKa value of the acid/base group cycles between high and low values during catalysis to optimize it for its role at each step of catalysis [5].
Enzymes of glycoside hydrolase families 18, 20, 56 and 84 hydrolyse substrates containing an N-acetyl (acetamido) group at the 2-position. These enzymes have no catalytic nucleophile: rather they utilize a mechanism in which the 2-acetamido group acts as an intramolecular nucleophile. Neighboring group participation by the 2-acdetamido group leads to formation of an oxazoline (or more strictly an oxazolinium ion) intermediate. This mechanism was deduced from X-ray structures of complexes of chitinases with natural inhibitors [6], from the potent inhibition afforded by a stable thiazoline analogue of the oxazoline [7], and from detailed mechanistic analyses using substrates of modified reactivity. Typically, a stabilizing residue (a carboxylate) stabilizes the charge development in the transition state. Not all enzymes that cleave substrates possessing a 2-acetamido group utilize a neighboring groups participation mechanism; enzyme of glycoside hydrolase families 3, 19 and 85 utilize a classical retaining mechanism with an enzymic nucleophile. Other hexosaminidases utilize an inverting mechanism.
References
- Gebler J, Gilkes NR, Claeyssens M, Wilson DB, Béguin P, Wakarchuk WW, Kilburn DG, Miller RC Jr, Warren RA, and Withers SG. (1992). Stereoselective hydrolysis catalyzed by related beta-1,4-glucanases and beta-1,4-xylanases. J Biol Chem. 1992;267(18):12559-61. | Google Books | Open Library
- Henrissat B, Callebaut I, Fabrega S, Lehn P, Mornon JP, and Davies G. (1995). Conserved catalytic machinery and the prediction of a common fold for several families of glycosyl hydrolases. Proc Natl Acad Sci U S A. 1995;92(15):7090-4. DOI:10.1073/pnas.92.15.7090 |
-
Koshland, D. (1953) Biol. Rev. 28, 416.
- McCarter JD and Withers SG. (1994). Mechanisms of enzymatic glycoside hydrolysis. Curr Opin Struct Biol. 1994;4(6):885-92. DOI:10.1016/0959-440x(94)90271-2 |
- McIntosh LP, Hand G, Johnson PE, Joshi MD, Körner M, Plesniak LA, Ziser L, Wakarchuk WW, and Withers SG. (1996). The pKa of the general acid/base carboxyl group of a glycosidase cycles during catalysis: a 13C-NMR study of bacillus circulans xylanase. Biochemistry. 1996;35(31):9958-66. DOI:10.1021/bi9613234 |
-
pmid= 7495789
-
pmid= 11124970