CAZypedia celebrates the life of Senior Curator Emeritus Harry Gilbert, a true giant in the field, who passed away in September 2025.
CAZypedia needs your help!
We have many unassigned pages in need of Authors and Responsible Curators. See a page that's out-of-date and just needs a touch-up? - You are also welcome to become a CAZypedian. Here's how.
Scientists at all career stages, including students, are welcome to contribute.
Learn more about CAZypedia's misson here and in this article. Totally new to the CAZy classification? Read this first.
Glycoside Hydrolase Family 82
This page has been approved by the Responsible Curator as essentially complete. CAZypedia is a living document, so further improvement of this page is still possible. If you would like to suggest an addition or correction, please contact the page's Responsible Curator directly by e-mail.
| Glycoside Hydrolase Family GH82 | |
| Clan | none |
| Mechanism | inverting |
| Active site residues | known |
| CAZy DB link | |
| https://www.cazy.org/GH82.html | |
Substrate specificities
All characterised members of glycoside hydrolase family 82 enzymes cleave the β-1,4 galactosidic bond of the marine algal polysaccharide iota-carrageenan [1, 2, 3] yielding products of the neocarrabiose series.
Kinetics and Mechanism
Family 82 enzymes operate with an inverting mechanism, as first shown by NMR [1] on the iota-carrageenase from Alteromonas fortis. The reaction seems to involve a chloride ion which through the Glu222 participated in the polarisation of the catalytic water molecule [2].
Catalytic Residues
From structural analysis the catalytic residues have been predicted to be two out of the three candidate amino acids Glu245, Asp247 or Glu310 in the A. fortis iota-carrageenase [4]. A study in 2010, which utilized site directed mutagenesis, has confirmed that Glu245 plays the role of the general acid residue in this inverting enzyme, and Asp247 is the general base activating the nucleophilic water molecule [2]. However, intriguingly the position of equivalent residues to Asp247 in other iota-carrageenase sequences are not strictly conserved [2, 3].
Three-dimensional structures
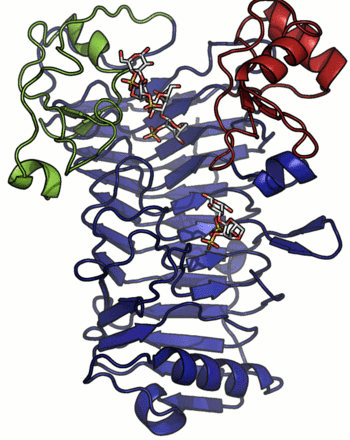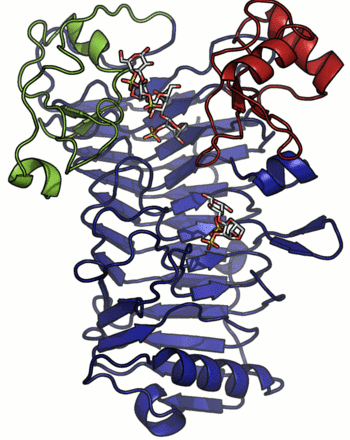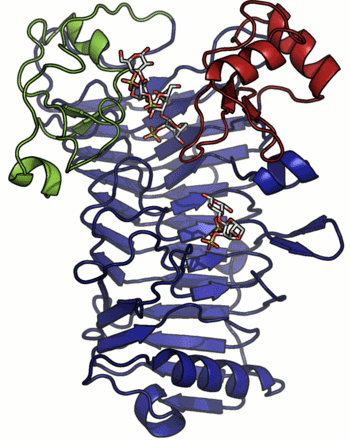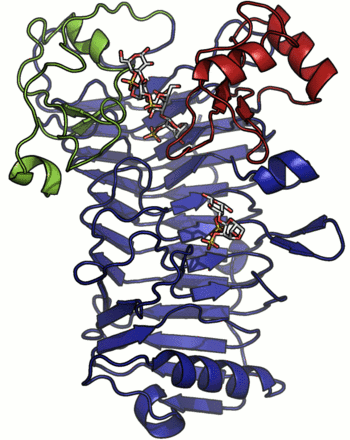
A crystal structure has only been determined for the iota-carrageenase from A. fortis and the protein folds into a right-handed parallel β-helix of 10 complete turns with two additional C-terminal domains (A and B) [4]. The crystal structure of a product complex has shed light on the existence of domain movement of domain A that is closed around the oligo-carrageenan in the complexed form and open in the uncomplexed enzyme (Figure 1) [5]. Recent discovery of shorter sequences indicates the existence of enzymes devoid of the C-terminal domain A or both domains A and B [2].
Family Firsts
- First sequence identification and family creation
- iota-carrageenase sequences have been first reported for enzymes from A. fortis and Z. galactanivorans [1].
- First sterochemistry determination
- GH82 enzymes are inverting as shown by NMR [1].
- First general acid residue identification
- Glu245 in the iota-carrageenase from Alteromonas fortis by site-directed mutagenesis and kinetic analysis [2]
- First general base residue identification
- Asp247 in the iota-carrageenase from Alteromonas fortis by site-directed mutagenesis and kinetic analysis [2].
- First 3-D structure
- iota-carrageenase from A. fortis [4]. The structure belongs to the β-helix fold (PDB 1h80 and PDB 1ktw).
References
- Barbeyron T, Michel G, Potin P, Henrissat B, and Kloareg B. (2000). iota-Carrageenases constitute a novel family of glycoside hydrolases, unrelated to that of kappa-carrageenases. J Biol Chem. 2000;275(45):35499-505. DOI:10.1074/jbc.M003404200 |
- Rebuffet E, Barbeyron T, Jeudy A, Jam M, Czjzek M, and Michel G. (2010). Identification of catalytic residues and mechanistic analysis of family GH82 iota-carrageenases. Biochemistry. 2010;49(35):7590-9. DOI:10.1021/bi1003475 |
- Hatada Y, Mizuno M, Li Z, and Ohta Y. (2011). Hyper-production and characterization of the ι-carrageenase useful for ι-carrageenan oligosaccharide production from a deep-sea bacterium, Microbulbifer thermotolerans JAMB-A94T, and insight into the unusual catalytic mechanism. Mar Biotechnol (NY). 2011;13(3):411-22. DOI:10.1007/s10126-010-9312-0 |
- Michel G, Chantalat L, Fanchon E, Henrissat B, Kloareg B, and Dideberg O. (2001). The iota-carrageenase of Alteromonas fortis. A beta-helix fold-containing enzyme for the degradation of a highly polyanionic polysaccharide. J Biol Chem. 2001;276(43):40202-9. DOI:10.1074/jbc.M100670200 |
- Michel G, Helbert W, Kahn R, Dideberg O, and Kloareg B. (2003). The structural bases of the processive degradation of iota-carrageenan, a main cell wall polysaccharide of red algae. J Mol Biol. 2003;334(3):421-33. DOI:10.1016/j.jmb.2003.09.056 |