CAZypedia celebrates the life of Senior Curator Emeritus Harry Gilbert, a true giant in the field, who passed away in September 2025.
CAZypedia needs your help!
We have many unassigned pages in need of Authors and Responsible Curators. See a page that's out-of-date and just needs a touch-up? - You are also welcome to become a CAZypedian. Here's how.
Scientists at all career stages, including students, are welcome to contribute.
Learn more about CAZypedia's misson here and in this article. Totally new to the CAZy classification? Read this first.
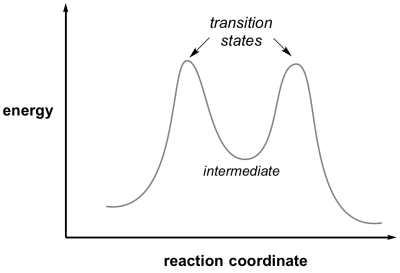
Reaction intermediate
This page has been approved by the Responsible Curator as essentially complete. CAZypedia is a living document, so further improvement of this page is still possible. If you would like to suggest an addition or correction, please contact the page's Responsible Curator directly by e-mail.
- Author: Stephen Withers
- Responsible Curator: Spencer Williams
An intermediate in a reaction is a species formed that has a real lifetime, best defined as being greater than the time for a bond vibration (approx 10-14 sec, ie a few femtoseconds). This will correspond to a minimum in the reaction coordinate diagram located between the reactant and product. Depending on their lifetimes and the tools available, intermediates can sometimes be trapped and observed by physical and chemical techniques.