CAZypedia needs your help! We have many unassigned GH, PL, CE, AA, GT, and CBM pages in need of Authors and Responsible Curators.
Scientists at all career stages, including students, are welcome to contribute to CAZypedia. Read more here, and in the 10th anniversary article in Glycobiology.
New to the CAZy classification? Read this first.
*
Consider attending the 15th Carbohydrate Bioengineering Meeting in Ghent, 5-8 May 2024.
Polysaccharide Lyase Family 7
This page is currently under construction. This means that the Responsible Curator has deemed that the page's content is not quite up to CAZypedia's standards for full public consumption. All information should be considered to be under revision and may be subject to major changes.
- Authors: ^^^Nadine Gerlach^^^ and ^^^Jan-Hendrik Hehemann^^^
- Responsible Curator: ^^^Jan-Hendrik Hehemann^^^
| Polysaccharide Lyase Family PL7 | |
| 3D Structure | β jelly roll |
| Mechanism | β-elimination |
| Active site residues | known |
| CAZy DB link | |
| http://www.cazy.org/PL7.html | |
Mechanism
Alginate lyases (Alys) of all families, PL5-7, PL14-15, PL17-18, cleave the glycosidic bond via β-elimination. Most PL7s are endo-active, i.e. acting within a poly- or oligosaccharide and releasing smaller alginate fragments. exo activity [1].
3 steps: [2] In both activity modes, a new nonreducing end with a 4-deoxy-L-erythro-hex-4 en pyranosyl uronate residue (Δ) is formed (Figure 1). In contrast to terrestrial PL7, marine PLs need the divalent cation calcium for substrate recognition and binding [1]. Ca2+ is weakening the ionic interactions between substrate (polyanion) and PL7 (polycation) by reducing the surface density of the alginate charge and therefore increasing the enzyme activity [3].
Kinetics and catalytic residues
Structural and biochemical analyses of wild type and mutated PL7s revealed five residues which are forming the active site: arginine (R), glutamine (Q), histidine (H), tyrosine (Y) [5], which are assmebled in three highly conserved regions (Figure 2). Histidine removes the negative charge of alginate, while
However, there can also be additional charged residues at the active site, which promote substrate recoginition and binding [1]. such residues can be found in the N-terminal R*ELREML and VIIGQIH regions. Both highly conserved regions were mainly characterized by hydrophobic amino acids (especially aromatic amino acids) such as leucine, tryptophan and methionine as well as residues with planar polar side chains (especially amino acids with charged side chains) such as arginine, glutamic acid, glutamine (Figure 2). These residues have been suggested to be substrate-binding molecules [6].
Substrate specificities
The polysaccharide lyase family 7 (PL7) contains five subfamilies (SF) based on their sequence similiarities [7], plus a so far uncharacterized sixth subfamily, which sonstist only of marine representatives of the Flavobacteriaceae [1]. All characterized PL7 enzymes were alginate lyases specific for the anionic, gel forming polysaccharide alginate. The substrate specificity depends on the source of alginate, i.e. derived from brown seaweed or mucoid bacteria Pseudomonas spp. and Azotobacter vinelandii, as well as geographical and saisonal parameters. Alginate is an heteropolysaccharide, consisting of β-D-mannuronate (M) and α-L-guluronate (G). These monosaccharides can occur in homogenous and heterogenous blocks. Hence, PL7 lyases can be mannuronate (EC 4.2.2.3), guluronate (EC 4.2.2.11) or mixed link (EC 4.2.2.-) specific. Despite the preference for M- or G-enriched blocks, most PL7s also have a moderate to low processivity for the other building block [1], [8].
SF3 & SF5 G-specific [1] [9, 10]
Substitution of hydrophobic amino acids in the isoleucine site of domain QIH could have an enormous influence on the high-affinity to pM or pG. This isoleucine was reconfirmed to be indispensable for recognition of the pG or G-G bond [11]
Three-dimensional structures
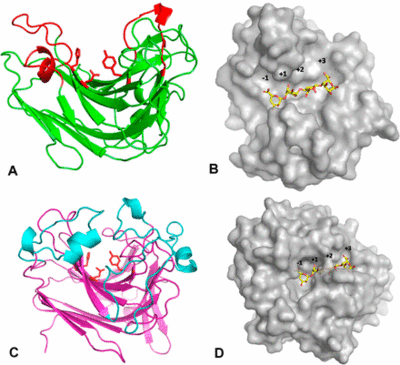
PL 7 is a very well biochemical characterized family with almost 40 entries in the CAZy data base [13]. Structural insights on the other hand are still restricted with nine 3D structures from only eight bacterial strains (status at CAZy in August 2019). The first structure of a PL7 was determined from Pseudomonas aeruginosa by multiple isomorphous replacement (MIR) at 2.0 Å resolution [14]. Just like PL14, PL7 belongs to the jelly roll family with a wide open cleft harboring the active site (Figure 3A, B). Til date, there is only one known exoactive PL7 structure [1].. Zobellia galaactinovorans DsijT is harboring, among others, two PL7 with two completely different activity modifs. AlyA1 belongs to SF3 and is an endo-active PL7. AlyA5 on the other hand belongs to SF5 and is exo-active, which active site is close by three additional loops forming a small pocket (Figure 3C, D).
The highly conserved 9-amino-acid-block YFKAGVY*Q, where * is a variable residue, at the C-terminus (Figure 2) has also been found for an extracellular pectate lyase in E. chrysanthemi (Keen & Tamaki, 1986) . Alginate and pectate / pectin lyases share several features such as β-elimination, the recognition of substrates of a similar structure and primary sequence similarity, indicating that they probably share a similar core structural fold. Since this conserved region was predicted in the cleft of the active site of PL7-1 despite having differing substrate specificity, it is likely not related to substrate recognition, but rather to maintaining a stable 3D-conformation [6].
CBM32: PDB:5ZU6 [15], [16] CBM16: unclear function [16] CBM13: [17]
Evolution of Aly PULs
lyases play different roles and have complementary activities [1] [18]
Family Firsts
- First catalytic endo-activity
- First catalytic exo-activity
- AlyA5 from Zobellia galactanivorans DsijT [1]
- First 3-D apo-structure
- PA1167 from Pseudomonas aeruginosa [14]
- First 3-D holo-structure
- A1-II from Sphingomons sp. A1 [12]
References
- Thomas F, Lundqvist LC, Jam M, Jeudy A, Barbeyron T, Sandström C, Michel G, and Czjzek M. (2013). Comparative characterization of two marine alginate lyases from Zobellia galactanivorans reveals distinct modes of action and exquisite adaptation to their natural substrate. J Biol Chem. 2013;288(32):23021-37. DOI:10.1074/jbc.M113.467217 |
- Favorov VV, Vozhova EI, Denisenko VA, and Elyakova LA. (1979). A study of the reaction catalysed by alginate lyase VI from the sea mollusc, Littorina sp. Biochim Biophys Acta. 1979;569(2):259-66. DOI:10.1016/0005-2744(79)90061-5 |
- Yamasaki M, Ogura K, Hashimoto W, Mikami B, and Murata K. (2005). A structural basis for depolymerization of alginate by polysaccharide lyase family-7. J Mol Biol. 2005;352(1):11-21. DOI:10.1016/j.jmb.2005.06.075 |
- Wong TY, Preston LA, and Schiller NL. (2000). ALGINATE LYASE: review of major sources and enzyme characteristics, structure-function analysis, biological roles, and applications. Annu Rev Microbiol. 2000;54:289-340. DOI:10.1146/annurev.micro.54.1.289 |
- Lombard V, Bernard T, Rancurel C, Brumer H, Coutinho PM, and Henrissat B. (2010). A hierarchical classification of polysaccharide lyases for glycogenomics. Biochem J. 2010;432(3):437-44. DOI:10.1042/BJ20101185 |
- Badur AH, Jagtap SS, Yalamanchili G, Lee JK, Zhao H, and Rao CV. (2015). Alginate lyases from alginate-degrading Vibrio splendidus 12B01 are endolytic. Appl Environ Microbiol. 2015;81(5):1865-73. DOI:10.1128/AEM.03460-14 |
-
&Yin2015 pmid=25831216
- Deng S, Ye J, Xu Q, and Zhang H. (2014). Structural and functional studies on three alginate lyases from Vibrio alginolyticus. Protein Pept Lett. 2014;21(2):179-87. DOI:10.2174/09298665113206660094 |
- Lombard V, Golaconda Ramulu H, Drula E, Coutinho PM, and Henrissat B. (2014). The carbohydrate-active enzymes database (CAZy) in 2013. Nucleic Acids Res. 2014;42(Database issue):D490-5. DOI:10.1093/nar/gkt1178 |
- Yamasaki M, Moriwaki S, Miyake O, Hashimoto W, Murata K, and Mikami B. (2004). Structure and function of a hypothetical Pseudomonas aeruginosa protein PA1167 classified into family PL-7: a novel alginate lyase with a beta-sandwich fold. J Biol Chem. 2004;279(30):31863-72. DOI:10.1074/jbc.M402466200 |
- Lyu Q, Zhang K, Zhu Q, Li Z, Liu Y, Fitzek E, Yohe T, Zhao L, Li W, Liu T, Yin Y, and Liu W. (2018). Structural and biochemical characterization of a multidomain alginate lyase reveals a novel role of CBM32 in CAZymes. Biochim Biophys Acta Gen Subj. 2018;1862(9):1862-1869. DOI:10.1016/j.bbagen.2018.05.024 |
- Sim PF, Furusawa G, and Teh AH. (2017). Functional and Structural Studies of a Multidomain Alginate Lyase from Persicobacter sp. CCB-QB2. Sci Rep. 2017;7(1):13656. DOI:10.1038/s41598-017-13288-1 |
- Li S, Yang X, Bao M, Wu Y, Yu W, and Han F. (2015). Family 13 carbohydrate-binding module of alginate lyase from Agarivorans sp. L11 enhances its catalytic efficiency and thermostability, and alters its substrate preference and product distribution. FEMS Microbiol Lett. 2015;362(10). DOI:10.1093/femsle/fnv054 |
- Hehemann JH, Correc G, Barbeyron T, Helbert W, Czjzek M, and Michel G. (2010). Transfer of carbohydrate-active enzymes from marine bacteria to Japanese gut microbiota. Nature. 2010;464(7290):908-12. DOI:10.1038/nature08937 |