CAZypedia needs your help! We have many unassigned GH, PL, CE, AA, GT, and CBM pages in need of Authors and Responsible Curators.
Scientists at all career stages, including students, are welcome to contribute to CAZypedia. Read more here, and in the 10th anniversary article in Glycobiology.
New to the CAZy classification? Read this first.
*
Consider attending the 15th Carbohydrate Bioengineering Meeting in Ghent, 5-8 May 2024.
Carbohydrate Binding Module Family 8
This page has been approved by the Responsible Curator as essentially complete. CAZypedia is a living document, so further improvement of this page is still possible. If you would like to suggest an addition or correction, please contact the page's Responsible Curator directly by e-mail.
| CAZy DB link | |
| http://www.cazy.org/CBM8.html |
Ligand specificities
These CBMs were originally called CBDVIII (family 8 cellulose-binding domains). DdCBM8, from the slime mold Dictyostelium discoideum CelA enzyme (270-6) binds xyloglucan, glucomannan, β-glucan, hydroxyethyl cellulose (HEC) and insoluble forms of cellulose, but not xylan [1]. Binding was detected using affinity gel electrophoresis, ITC and intrinsic fluorescence microscopy [1]. There is no evidence for binding to oligosaccharides [1].
Structural Features
DdCBM8 is found C-terminal to the GH9 endo-(1,4)-beta-D-glucanase (cellulase) catalytic module of the CelA enzyme (270–6) and connected via a Thr-Glu-Thr-Pro type repeat linker [1, 2].
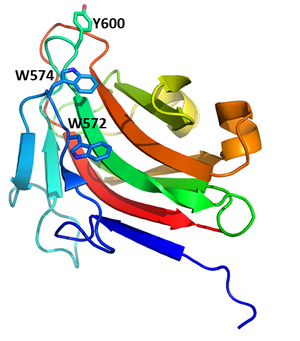
DdCBM8 has a beta-sandwich fold which supports the planar surface formed by W572, W574, and Y600, akin to type A CBMs [1]. These residues align with the CBM29-2 1GWM binding site [3], the residues R634 and Q686 from DdCBM8 are also conserved with binding residues from CBM29-2 [1]. Alanine mutations were made in DdCBM8 for W572, Y600, F608, R634, and Q686 and glycan binding was assessed using affinity gel electrophoresis [1]. The mutations W572A and Y600A abolished binding. The researchers were unable to produce the protein for the mutation W574A [1]. No shift in gel mobility was detected for F608A or R634A, though the mutation Q686A did have an effect confirming its role in ligand binding [1]. The W572,W574, Y600, and Q686 residues are highly conserved in the CBM8 family [1].
DdCBM8 appears to be broadly specific with both type A and type B CBM characteristics as it binds crystalline cellulose with a planar binding site, but has higher affinity for soluble glycans [1].
Functionalities
DdCBM8 is appended to the GH9 endo-(1,4)-beta-D-glucanase CelA [2]. DdCBM8 shows fairly promiscuous binding so it may bring the enzyme in proximity to substrate (it is unlikely target to specific motifs).
CelA and CelB (a cellulose binding domain with no known cellulase activity [4]) from Dictyostelium discoideum are predicted to be important for amoebae release from spores as transcriptomic experiments show that their mRNA levels are low in dormant spores, they rise during germination and then rapidly disappear after germination [2]. Cellulase activities from differently sized cellulases are also shown to increase during spore germination [5]. It is suggested that CelB might improve the substrate accessibility for CelA due to their concomitant expression during germination [4].
Family Firsts
- First Identified
- The first suggestion of binding to cellulose for the CBM8 family was determined by affinity gels in the absence of the DdCBM8 from CelA from Dictyostelium discoideum [2]. Detailed binding studies are presented in ref. [1].
- First Structural Characterization
- The first crystal structures are from DdCBM8, from the slime mold Dictyostelium discoideum [1], see 7T7Y and 7T7Z.
References
- Liberato MV, Campos BM, Tomazetto G, Crouch LI, Garcia W, Zeri ACM, Bolam DN, and Squina FM. (2022). Unique properties of a Dictyostelium discoideum carbohydrate-binding module expand our understanding of CBM-ligand interactions. J Biol Chem. 2022;298(5):101891. DOI:10.1016/j.jbc.2022.101891 |
- Ramalingam R, Blume JE, and Ennis HL. (1992). The Dictyostelium discoideum spore germination-specific cellulase is organized into functional domains. J Bacteriol. 1992;174(23):7834-7. DOI:10.1128/jb.174.23.7834-7837.1992 |
- Charnock SJ, Bolam DN, Nurizzo D, Szabó L, McKie VA, Gilbert HJ, and Davies GJ. (2002). Promiscuity in ligand-binding: The three-dimensional structure of a Piromyces carbohydrate-binding module, CBM29-2, in complex with cello- and mannohexaose. Proc Natl Acad Sci U S A. 2002;99(22):14077-82. DOI:10.1073/pnas.212516199 |
- Ramalingam R and Ennis HL. (1997). Characterization of the Dictyostelium discoideum cellulose-binding protein CelB and regulation of gene expression. J Biol Chem. 1997;272(42):26166-72. DOI:10.1074/jbc.272.42.26166 |
- Blume JE and Ennis HL. (1991). A Dictyostelium discoideum cellulase is a member of a spore germination-specific gene family. J Biol Chem. 1991;266(23):15432-7. | Google Books | Open Library
- Tomme P, Boraston A, McLean B, Kormos J, Creagh AL, Sturch K, Gilkes NR, Haynes CA, Warren RA, and Kilburn DG. (1998). Characterization and affinity applications of cellulose-binding domains. J Chromatogr B Biomed Sci Appl. 1998;715(1):283-96. DOI:10.1016/s0378-4347(98)00053-x |
-
P. Tomme, R.A.J. Warren, R.C. Miller Jr., D.G. Kilburn, N.R. Gilkes, in: J.N. Saddler, M.H. Penner (Eds.), Enzymatic Degradation of Insoluble Carbohydrates, American Chemical Society Symposium Series, 1995, p. 142.